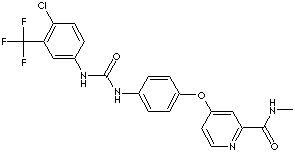
PRODUCT IDENTIFICATION
284461-73-0 (parent)
475207-59-1 (tosylate)
H.S. CODE
TOXICITY
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
white to off-white powder
REFRACTIVE INDEX
NFPA RATINGS
AUTOIGNITION
FLASH POINT
GENERAL DESCRIPTION & EXTERNAL LINKS
Types of chemotherapy drug;
- Alkylating agents
- Angiogenesis Inhibitors
- Anti-metabolites
- Anti-mitotics
- Aromatase inhibitors
- Biologic response modifiers
- Differentiating agents
- Hormonal therapies
- Monoclonal antibodies
- Platinum derivatives
- Signaling inhibitors
- Topoisomerase inhibitors
Treating the cancer by enhancing the immune system's ability to destroy it causes some cancers to shrink and may prolong survival (see Prevention and Treatment of Cancer: Immunotherapy). One such treatment, interleukin-2, is used for kidney cancer. Various combinations of interleukin-2, interferon, and other biologic agents and even vaccines developed from cells removed from the kidney cancer are being investigated. These treatments may be helpful for metastatic cancer, although the benefit is usually small. For people with metastatic cancer, recently developed treatments include the drugs sunitinib, sorafenib, and temsirolimus. These drugs alter molecular pathways that affect the tumor and are thus called targeted therapies. Rarely (in less than 1% of people), removing the affected kidney causes tumors elsewhere in the body to shrink. However, the slim possibility that tumor shrinkage will occur is not considered sufficient reason to remove a cancerous kidney when the cancer has already spread, unless removal is part of an overall plan that includes other systemic therapies.(http://www.mercksource.com/)
Sorafenib may stop the growth of tumor cells by blocking blood flow to the tumor and by blocking some of the enzymes needed for cell growth. Estradiol can cause the growth of breast cancer. Hormone therapy using anastrozole may fight breast cancer by blocking the use of estradiol by the tumor cells. Sometimes when hormone therapy is given, it does not stop the growth of tumor cells. The tumor is said to be resistant to hormone therapy. Giving sorafenib together with anastrozole may reduce drug resistance and allow the tumor cells to be killed. (http://www.clinicaltrials.gov/show/NCT00217399)
Sorafenib (Nexavar®) and sunitinib (Sutent®) are orally administered inhibitors of protein tyrosine kinases associated with the intracellular portions of certain transmembrane receptor molecules. Each targets the tyrosine kinase activity of more than one receptor, with effects that depend on each cell type’s receptor repertoire. Inhibiting these intracellular tyrosine kinases blocks signal transduction after a growth factor, cytokine, or other ligand binds to the receptor’s extracellular domain. In different cell types, this can inhibit tumor growth, metastasis, or angiogenesis (growth of new blood vessels). Both sorafenib and sunitinib are approved by the U.S. Food and Drug Administration (FDA) for the treatment of advanced renal cell carcinoma. Sunitinib also is indicated to treat gastrointestinal stromal tumor (GIST) after disease progression on or intolerance to imatinib. (http://www.bcbs.com/)
|
|
APPEARANCE
white to off-white powder
IDENTIFICATION
to pass
ASSAY
99.0%
HEAVY METALS
10ppm max
LOSS ON DRYING
0.5% max
0.2% max
TOTAL IMPURITY
1.0% max